Conversations with Industry: Fisher & Paykel Healthcare
This content is written by the company and does not represent the opinions of ACEP.
Nasal High Flow and COVID-19 Respiratory Illness
Research and experience from UnityPoint Health.
Preventing Nosocomial Transmission Recommendations
Research from the International Society of Aerosol Medicine
NHF implementation with COVID-19 patients
Perspective
Guidelines for the clinical management of COVID-19 from organizations, such as the NIH, SSC, and ACEP, recommend the use of Nasal High Flow as respiratory support in adults. These recommendations for the use of Nasal High Flow are supported by analyzed data from research investigating the effects of Nasal High Flow on clinical outcomes, such as the reduced need for intubation.
Nasal High Flow was implemented on COVID-19 patients in the U.S. hospital, UnityPoint Health, based on their clinical research. Part of their protocol is outlined below.
Recommendations:
- Nasal High Flow Therapy has a role in a subset of COVID-19 patients who have normal lung compliance but have severe symptomatic hypoxemia.
- The pulmonary/critical care providers will be required to have oversight of management of patients on NHF who are highly suspect of or confirmed with COVID-19.
- If another provider wants to use NHF, pulmonary/critical care will be consulted and make the ultimate decision on appropriateness of oxygen therapy and patient placement.
- The World Health Organization (WHO) does not consider Nasal High Flow as aerosol-generating. Their guidelines support the use of Nasal High Flow oxygen in some patients, but they urge close monitoring for clinical deterioration that could result in the need for emergent intubations.
Indications for Use:
- Use in Patients
- P/F > 150
- Patients that are requiring more than 4L of oxygen via NC or FiO2 ≥ 0.5 and have adequate Respiratory Rate
- Patients with asymptomatic hypoxemia and evidence of end organ or tissue hypoxia
- Support of hypoxemic patients who are DNR/DNI and being treated with anti-viral medication and need to survive two days to determine if medication will work)
- Do not use in air hunger in patients on comfort care. Provide them with an NRB mask if needed.
Infection Control Safety:
- Droplet + contact + eye protection precautions if within 6 feet of patient while on the device.
- Droplet + contact isolation room procedures.
Safety:
- Patient should have surgical mask placed over face and cannula when staff and providers are within 6 feet of the patient for care.
- Turn off NHF every time before removing the device from patient’s face.
- Correct size with snug fit of interface
- Patient should not be transported while on the device. Transport should be done with NRB mask tightly secured to patient face.
- Patient should be moved to the ICU for closer monitoring and care if requiring more than 50 liters or FiO2 ≥ 50%. Exception is if patient is DNR/DNI and no further escalation will be required.
- Pulmonary/critical care providers have the final discretion to accept patient to the Intensive care unit. If not accepting, documentation will be noted in the electronic medical record (EMR).
FAQ: Nasal High Flow as Respiratory Support in Adults
What is an example of real-world use and implications of using Nasal High Flow with COVID-19 patients?
Watch this recent news story (video below) highlighting the real-world use and implications of using Nasal High Flow with COVID-19 patients. This news report shows how UnityPoint Health implemented Nasal High Flow to reduce the need for intubation, based on their clinical research and expert body guidelines.
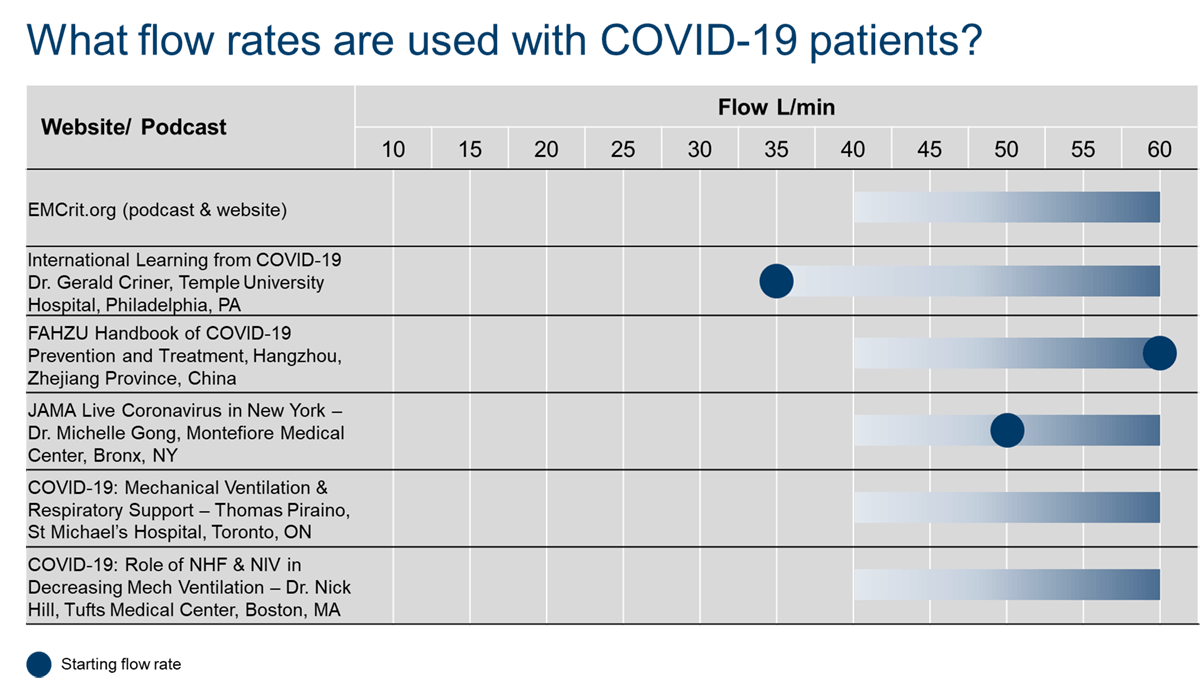
What flow rates should be used with Nasal High Flow for COVID-19 patients, according to expert guidelines?
Want to start a conversation with us? We love to hear from you.



