
Toxic Alcohols
A 33yo male presents to the emergency room with suicidal ideation. He describes that he drank “a few sips” of windshield washer fluid about half an hour ago. The patient’s vitals are T: 98.4F, HR: 84, BP: 132/85, RR: 14, O2sat: 100% on RA. You verify the brand of windshield fluid as “Rain-X De-icer.” You consult an MSDS and find that it contains “Methyl Alcohol.” Labs are shown below.

Serum osmolality: 340 mOsm/kg
ETOH: < 10
APAP: < 5
Salicylate: < 3
Beta-hydroxybutyrate: 0.03 mmol/L
Introduction
Toxic alcohols are simply a way to refer to organic compounds that contain one or more hydroxyl groups (-OH) that are not designated for human consumption. The most common and well-known toxic alcohols are methanol, ethylene glycol and isopropanol.
Methanol (methyl alcohol, “wood alcohol”). It is a colorless, volatile, flammable liquid that can smell like ethanol. It is commonly found in deicer fluid, cooking fuel, carburetor fluid and as a contaminant in moonshine. Ethylene glycol is an odorless, colorless, viscous liquid. It has a sweet taste, which increases the risk of significant accidental ingestions. It is used in industry as a reagent and as the main component in anti-freeze. Isopropanol is also known colloquially as “rubbing alcohol.” It is mainly found in disinfectants, used as a solvent in industry and finds use in laboratory applications.
The main route of exposure is through ingestion and absorption through the GI tract, which has near 100% bioavailability for all three toxic alcohols. Exposure can occur via inhalation for more volatile alcohols, such as methanol and isopropanol, but toxicity is rare. In certain instances, dermal exposure, particularly chronic exposure, can result in toxicity. This route of exposure has become important in hand sanitizer containing methanol[2,3,4].
Metabolism and Pathophysio
When it comes to understanding why toxic alcohols are poisonous, we must look at their metabolism. Beyond intoxication and CNS depression, toxic alcohols in of themselves do not possess significant toxicity. The toxicity of these compounds comes from their organic acid metabolites. They result in the hallmark anion gap metabolic acidosis while also having unique end organ effects.
The two important enzymes involved are alcohol dehydrogenase (ADH) and aldehyde dehydrogenase (ALDH). These enzymes function by oxidizing the parent molecule; this is coupled to a reduction in NAD+ to form NADH.
Methanol is oxidized to formaldehyde by ADH, which is then oxidized to formic acid by ALDH. Formic acid (undissociated) exists in an equilibrium with formate (dissociated). Formic acid is the main toxic metabolite. Formate can be further metabolized to CO2 and water when it combines with folate, providing a pathway for formic acid to be neutralized to non-toxic metabolites. Very little is expired and excreted in sweat, unchanged.
Ethylene glycol is metabolized to glycoaldehyde by ADH then to glycolic acid by ALDH which then is converted to glyoxylic acid and oxalic acid. The primary toxic metabolites are glycolic acid and oxalic acid, although glyoxylic acid likely contributes to the anion gap metabolic acidosis. Glyoxylic acid can be metabolized by two other pathways, combining with thiamine to form α-hydroxy-β-ketoadipic acid and pyridoxine to form glycine, which is further metabolized to hippuric acid.
Isopropanol is the exception among these three toxic alcohols. It is metabolized by ADH to acetone. This is due to isopropanol being a secondary alcohol, which are metabolized to ketones by ADH. Because acetone is not an aldehyde, it is not able to be further metabolized. This leads to a ketosis. Because there is no resulting formation of organic acids, there is no acidosis. Hence the mantra of “Ketosis without acidosis.” Acetone is simply excreted by the kidneys.
Clinical effects
In general, patients who ingest a toxic alcohol will initially develop intoxication, similar to ethanol. These patients may also have generalized nausea, vomiting, and abdominal pain. Vital sign abnormalities can vary, but shortly after ingestion will often be normal.
The hallmark metabolic abnormality that is associated with toxic alcohols is the development of a metabolic acidosis due to the organic acid metabolites. The acidosis can lead to general organ dysfunction, such as kidney injury, rhabdomyolysis, CNS depression and cardiotoxicity. Some of these effects overlap with specific toxic effects of the alcohol or metabolites.
Methanol’s main claim to fame is visual impairment, which occurs on a spectrum ranging from blurry vision to complete blindness. The vision loss is often asymmetric[8,9]. The formic acid is responsible for toxicity, as it inhibits oxidative phosphorylation, preferentially affecting cells in the optic nerve. CNS toxicity results from toxicity to the basal ganglia, which is also found in carbon monoxide poisoning and hypoxia[4,10]. Other effects include acute kidney injury, liver injury, pancreatitis and rhabdomyolysis.
Ethylene glycol’s most prominent effect is nephrotoxicity due to precipitation of oxalic acid with calcium to form calcium oxalate crystals. Consequently, hypocalcemia can occur along with resultant dysrhythmias. CNS toxicity leads to cerebral edema, cranial nerve deficits and seizures. The underlying mechanism is thought to be due to calcium oxalate crystals precipitating in the CNS.
Isopropanol is associated with hemorrhagic gastritis due to its local irritant effect. Ingestions of isopropanol are rarely lethal.
Management
As with all patients, airway, breathing and circulation should be evaluated with potential life threats addressed. Respiratory failure or concerns for airway protection should be addressed appropriately. Hypotension should be addressed with IV fluids and vasopressors.
The mainstay of treatment for methanol and ethylene glycol is administration of fomepizole (4-methylpyrazole). This treatment should be given in any case where there is enough clinical suspicion for toxic alcohols, whether it is based on history indicating ingestion or lab abnormalities such as unexplained acidosis or presence of an osmolar gap. Fomepizole functions by inhibiting the action of alcohol dehydrogenase, preventing metabolism of toxic alcohols to their metabolites. This effectively prolongs the half-life of methanol to 54 hours[11], ethylene glycol[12] to 11-18 hours and isopropanol to 28 hours[13].
The dosing of fomepizole involves giving a loading dose of 15mg/kg over 30 minutes. This is followed by 10mg/kg every 12 hours for the first 48 hours. If a patient is to remain on fomepizole for longer than 48 hours, the dose must be increased to 15mg/kg to offset fomepizole’s ability to induce its own metabolism. Dosing must also be adjusted during dialysis (discussed further below)
In places without access to fomepizole, ethanol given as an infusion or orally can be used to block ADH. ADH has a much higher affinity for ethanol than other toxic alcohols, allowing ethanol to act as a competitive inhibitor. The main problem with ethanol infusion is that it requires close monitoring to maintain serum level of 100mg/dL. Oral administration can be used if intensive monitoring is not available, although titration with oral administration can be more difficult. Dosing is highly variable, with higher dosages required for more tolerant patients.
Adjunctive treatments for toxic alcohols can also be given to help with clearance of toxic metabolites and to prevent further toxic effects. Sodium bicarbonate is important in methanol poisoning because it promotes dissociation of formic acid to formate, which can then be metabolized to CO2 and water. An infusion should be started with a pH goal of > 7.2. Supplementation with folic acid or folinic acid (leucovorin) (1-2mg/kg max 50-70mg IV every 6 hours) promotes metabolism of formate. In ethylene glycol poisoning, metabolism of glyoxylic acid to less toxic metabolites is promoted by supplementation of thiamine (100mg q8hr in adults and 10-50mg q8hr in children) and pyridoxine (50mg IV/IM every 6 hours).
Toxic alcohols are readily removed by extracorporeal removal techniques (ECRT), such as hemodialysis and continuous renal replacement therapy (CRRT). ECRT is particularly important consideration for patients with methanol poisoning, as methanol’s half-life is significantly increased in the presence of fomepizole. Indications for dialysis as put forth by the EXTRIP guidelines can be found in table 1[14]. Dialysis decreases half-life of methanol to 2-3 hours[15,16] and ethylene glycol to 3-4 hours[17] . Depending on the patient’s serum concentration of methanol or ethylene glycol, they may require a prolonged session of dialysis or may require multiple sessions. ECRT should be considered for patients with ethylene glycol level > 50 mg/dL, severe acidosis, renal failure or CNS findings. Goal concentration after dialysis for both methanol and ethylene glycol is <20mg/dL. Hemodialysis also readily clears organic acid metabolites, preventing further damage to end organs.
In set the setting of dialysis, dosing of fomepizole must be adjusted before, during and after. Prior to the start of dialysis, if more than 6 hours have elapsed since the last dose, an additional dose should be given. During dialysis, manufacturer recommendation is giving one full dose every four hours the patient is on dialysis. After dialysis is completed, dosing is adjusted based when the most recent dose was given. If <1 hour, no additional dose is required. If 1-3 hours, 50% of the next scheduled dose and if more than 3 hours, a full dose should be given. Regular dosing can then resume every 12 hours.
Recommended Indications for dialysis for Methanol (EXTRIP)
- Presence of coma, seizures or new visual deficits
- Persistent acidosis despite adequate supportive measures or Blood pH ≤ 7.15
- Anion Gap > 24
- Serum methanol concentration > 70 mg/dL in context of fomepizole treatment
- Serum methanol concentration > 60 mg/dL in context of ethanol treatment
- Serum methanol concentration > 50 mg/dL in absence of ADH blocker
- Impaired renal function
Diagnostics
Once a patient is initially stabilized, patients with suspicion for toxic alcohol ingestion should have evaluation of their acid/base status and electrolytes with a CBC, CMP, magnesium, lactate, ABG/VBG, ETOH level, and beta-hydroxybutyrate. Serum methanol, ethylene glycol and isopropanol levels should also be obtained. Depending on your laboratory’s capability, these may be a send out test.
Early after ingestion, you may see an osmolar gap due to increase in unaccounted for osmols in the form of the toxic alcohol.
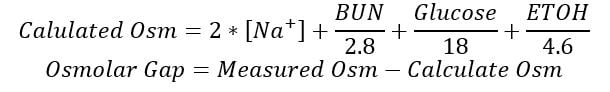
A normal osmolar gap is between -14 to +10 mOsm/L [18]. Toxic alcohols should be on your differential in the setting of an abnormal positive osmolar gap. It is important to realize that the lack of an osmolar gap does not preclude a toxic alcohol ingestion. This is due to the wide variability in “normal” osmolar gaps in the population and metabolism of toxic alcohols. There are numerous equations that are used to calculate the osmolar gap and each has its own reference range and there is individual variability. What could be normal in one person, may be indication of a toxic alcohol ingestion. Later in the time course of a toxic alcohol ingestion, patients will often have normal osmolar gaps as the original toxic alcohol has already been fully metabolized. This can be falsely reassuring and should not be used to rule out a toxic alcohol[19].
Shortly after ingestion, a patient will have a normal anion gap, as little toxic alcohol has been metabolized and there is very little organic acid present. As metabolism continues and the serum concentration of organic acids increases, the anion gap will also increase. Bicarbonate acts as a buffer in serum and will decrease in response to an increase in acid load. In the case of toxic alcohols, the organic acid metabolites are buffered initially by the bicarbonate in the serum. The reduction in bicarbonate concentration represents the increase in unmeasured organic acids. The unaccounted anions are formate in methanol and glycolate/glyoxylate/oxalate in ethylene glycol. A normal anion gap typically is < 12 mEq/L. Toxic alcohols should higher on the differential for patients who have an unexplained anion gap metabolic acidosis, even in the setting of a normal osmolar gap. The anion gap is calculated as follows.
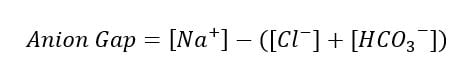
Both the osmolar gap and anion gap must be considered in the setting of a toxic alcohol ingestion. A decreasing osmolar gap with an increasing anion gap makes toxic alcohol poisoning more likely, but is not necessarily diagnostic.
The following additional diagnostic tests are neither sensitive nor specific for toxic alcohol poisoning, but may provide additional evidence to further support or rule out the diagnosis. A urinalysis can be helpful if calcium oxalate crystals are present or there is fluorescence under UV light, which may be suggestive of ethylene glycol ingestion. Note that the fluorescence is not due to the ethylene glycol itself, but rather a fluorescent dye added to anti-freeze that helps mechanics identify a leak in a car’s engine. In these patients where toxic alcohol poisoning is of concern, consider early Toxicology or Poison Control Center consultation to aid in management.
A false lactate elevation can be seen in ethylene glycol ingestions. This occurs if the method of measurement uses lactate oxidase to measure lactate. This is often found in point of care blood gas analyzers. This is because the glycolate has similar structure to lactate and is readily metabolized by lactate oxidase. The POC analyzer interprets a color change as a result of the reaction to calculate the lactate level. This will result in a measured lactate that appears high. True serum measurement as performed in most hospital laboratories using lactate dehydrogenase (more specific to lactate) will result in a true lactate. The difference between these two measurements, the “lactate gap,” can provide additional evidence for ethylene glycol ingestion[20].
There is a case report of methanol toxicity being mistaken for ethanol intoxication due to use of a breathalyzer, where the methanol in the breath was mistakenly detected as ethanol by the device leading to delayed diagnosis[21].
In the setting of isopropanol ingestion, the increase in serum acetone metabolites can lead to a falsely elevated creatinine. This is due to acetone’s interference with the Jaffe reaction, which is a common method of measurement for creatinine. You can still trend creatinine to determine worsening of kidney function, but the absolute value is difficult to interpret.
Case resolution
You recognize that methyl alcohol is also known as methanol. You order fomepizole to block the patient. You obtain an osmolar gap of 49.0 mOsm/kg. You also obtain methanol, ethylene glycol and isopropanol levels, which showed a methanol level of 130 mg/dL. You coordinate admission and dialysis with the medicine and nephrology teams and the patient is admitted. He does well and subsequently is discharged to inpatient psychiatry 2 days later.
|
|
Methanol |
Ethylene Glycol |
Isopropanol |
|
Potential routes of exposure |
Oral, inhalation, dermal |
Oral |
Oral, inhalation, dermal |
|
Anion Gap Metabolic Acidosis |
Yes |
Yes |
No |
|
Toxic metabolites |
Formic acid, formaldehyde |
Glycolic acid, Glyoxylic acid, oxalic acid |
Acetone |
|
Clinical effects |
CNS Depression Visual changes Liver injury Pancreatitis Rhabdomyolysis Seizure |
CNS Depression Nephrotoxicity Cranial Nerve palsies Cerebral edema Seizure
|
CNS Depression Hemorrhagic gastritis |
|
Adjunctive treatments |
Folate, sodium bicarbonate |
Thiamine, Pyridoxine |
None |
References:
- Nelson, Lewis S.; Howland, Mary Ann; Lewin, Neal A.; Smith, Silas W.; Goldfrank, Lewis R.; Hoffman, Robert S. Goldfrank's Toxicologic Emergencies, 11e. New York: McGraw-Hill, 2019.
- Adanir T, et al. Percutaneous methanol intoxication: case report. Eur J Anaesthesiol. 2005;22:560-561.
- Dogan H, Yilmaz Karakus B, Serefoglu Cabuk K, Uzun O, Yenice H, Orucoglu A. Transdermal Spirit (Methanol) Poisoning: A Case Report. Iran Red Crescent Med J. 2016;18(11):e23767. Published 2016 Jan 25. doi:10.5812/ircmj.23767
- Karaduman F, et al. Bilateral basal ganglionic lesions due to transdermal methanol intoxication. J Clin Neurosci. 2009;16:1504-1506.
- Ashurst JV, Nappe TM. Methanol Toxicity. [Updated 2020 Jun 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: Learn More
- Iqbal A, Glagola JJ, Nappe TM. Ethylene Glycol Toxicity. [Updated 2020 Jun 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: Learn More
- Ashurst JV, Nappe TM. Isopropanol Toxicity. [Updated 2020 Jun 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: Learn More
- Chung TN, et al. Unilateral blindness with third cranial nerve palsy and abnormal enhancement of extraocular muscles on magnetic resonance imaging of orbit after the ingestion of methanol. Emerg Med J.Emerg Med J. 2010;27:409-410.
- Lu JJ, et al. Unilateral blindness following acute methanol poisoning. J Med Toxicol. 2010;6:459-460.
- Sefidbakht S, et al. Methanol poisoning: acute MR and CT findings in nine patients. Neuroradiology. 2007;49:427-435.
- Brent J, McMartin K, Phillips S, Aaron C, Kulig K; Methylpyrazole for Toxic Alcohols Study Group. Fomepizole for the treatment of methanol poisoning. N Engl J Med. 2001;344(6):424-429. doi:10.1056/NEJM200102083440605
- Levine M, Curry SC, Ruha AM, et al. Ethylene glycol elimination kinetics and outcomes in patients managed without hemodialysis. Ann Emerg Med. 2012;59(6):527-531. doi:10.1016/j.annemergmed.2011.12.001
- Bekka R, Borron SW, Astier A, Sandouk P, Bismuth C, Baud FJ. Treatment of methanol and isopropanol poisoning with intravenous fomepizole. J Toxicol Clin Toxicol. 2001;39(1):59-67. doi:10.1081/clt-100102881
- Roberts DM, Yates C, Megarbane B, Winchester JF, Maclaren R, Gosselin S, Nolin TD, Lavergne V, Hoffman RS, Ghannoum M; EXTRIP workgroup. (2015) "Recommendations for the Role of Extracorporeal Treatments in the Management of Acute Methanol Poisoning: A Systematic Review and Consensus Statement." Critical Care Medicine 43(2): 461-472.
- Lachance P, Mac-Way F, Desmeules S, et al. Prediction and validation of hemodialysis duration in acute methanol poisoning. Kidney Int. 2015;88(5):1170-1177. doi:10.1038/ki.2015.232
- Kraut JA. Approach to the Treatment of Methanol Intoxication. Am J Kidney Dis. 2016;68(1):161-167. doi:10.1053/j.ajkd.2016.02.058
- Iliuta IA, Lachance P, Ghannoum M, et al. Prediction and validation of the duration of hemodialysis sessions for the treatment of acute ethylene glycol poisoning. Kidney Int. 2017;92(2):453-460. doi:10.1016/j.kint.2017.02.018
- Hoffman RS, Smilkstein MJ, Rowland MA, Goldfrank LR. Osmol gaps revisited: Normal values and limitations. J Toxicol Clin Toxicol 1993;31(1):81–93.
- Corcoran, J., 2020. Why Osms… aren’t so awesome. [Blog] Everything is Osm, Available at: <https://everythingisosm.com/2020/03/03/why-osms-arent-so-awesome/> [Accessed 21 July 2020].
- Sagar AS, Jimenez CA, Mckelvy BJ. Lactate gap as a tool in identifying ethylene glycol poisoning. BMJ Case Rep. 2018;2018:bcr2018224243. Published 2018 Mar 9. doi:10.1136/bcr-2018-224243
- Caravati EM, Anderson KT. Breath alcohol analyzer mistakes methanol poisoning for alcohol intoxication. Ann Emerg Med. 2010;55(2):198-200. doi:10.1016/j.annemergmed.2009.07.021
Richard Chen, MD
Medical Toxicology Fellow
Einstein Medical Center



