CAR-T Therapy and the Cytokine Storm

Among the recent advances in immunotherapy for cancer has been the development of chimeric antigen receptor T cells, or CAR-T cells. Despite remarkable efficacy in the treatment of some malignancies, CAR-T therapy has several notable adverse reactions which can be life threatening and require novel treatments not routinely utilized by the emergency physician/intensivist.
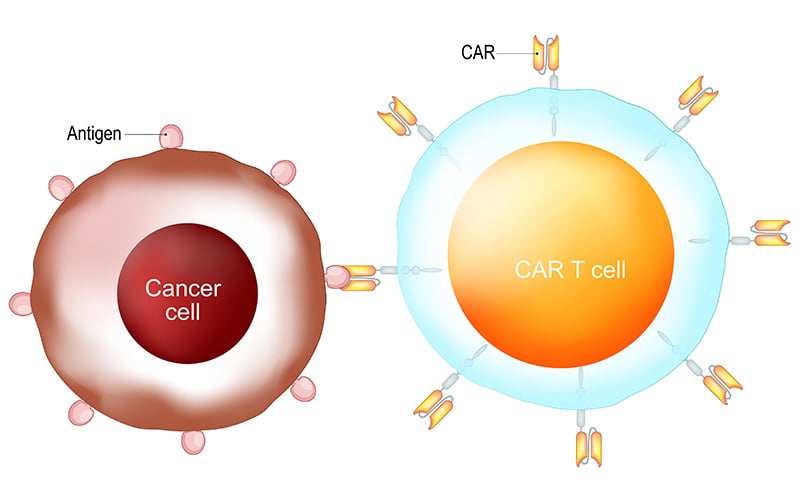
CAR-T cells are created by isolation of native T cells from the patient via leukapharesis. In a lab, the harvested T cells are infected with a virus encoding the chimeric antigen receptor to target the patient’s specific type of cancer. Once infected with the virus, the T cells display both the appropriate antigen receptor, as well as the costimulatory molecules required to activate the T cell against the targeted antigen. These T cells are clonally expanded and then re-infused into the patient after pretreatment chemotherapy.1
Currently, CAR-T therapy is only approved for the treatment of refractory B-cell ALL and large B-cell lymphoma – targeting the cell surface protein CD-19 – although many different targets are being studied in clinical trials.2
The most common severe reaction to CAR-T therapy is the cytokine release syndrome (CRS). CRS occurs after the hundreds of millions of infused T cells release cytokines in a positive feedback loop, causing a systematic inflammatory response syndrome (SIRS). The SIRS reaction present in CRS can be clinically indistinguishable from sepsis and septic shock, with fevers, tachycardia, hypotension, and multiple organ system dysfunction.
Over 75% of patients treated with CAR-T therapy will develop CRS and about 25-50% will develop a severe CRS, with the greatest risk factor being a high tumor burden. The onset is typically within a few days of the first infusion and takes about a week to resolve, on average.
The fundamentals of management of CRS are similar to those required for septic shock, including broad-spectrum antibiotics (as the patients are typically immunosuppressed), intravenous fluids and antipyretics, and vasopressors, as needed. However, in contrast to septic shock, the cytokines are directly targeted with an interleukin-6 antagonist, either tocilizumab (an IL-6 receptor antibody) or siltuximab (a direct IL-6 antibody). Multiple doses may be required, and the use does not appear to blunt efficacy. The decision to proceed with IL-6 therapy is typically made in conjunction with the treating oncologist. “Resistant” CRS, defined as worsening or lack of clinical improvement after IL-6 blockade, is treated with high dose corticosteroids, typically at least 2 mg/kg of methylprednisolone per day, or equivalent). In contrast to IL-6 blockade, administration of corticosteroids does appear to blunt the efficacy of the treatment but may be required in up to a quarter of patients treated with CAR-T therapy.3
In addition to CRS, neurologic toxicity is common with CAR-T therapy, seen in over 50% of patients. Typically occurring four-six days after infusion of the therapy, neurologic sequelae can be seen up to a year out from treatment and typically last one to two weeks. A wide variety of symptoms have been described; the most common symptoms are encephalopathy, headache, and tremor, but may include agitated delirium, focal weakness, or aphasia.4 IL-6 blockade appears to worsen these symptoms, so in the absence of concurrent CRS, symptoms are typically treated with corticosteroids.
CAR-T therapy is rapidly expanding to new indications and malignancies, and so the observed side effects are likely to expand in the coming years as further research is published. The emergency physician/intensivist is likely to be at the front lines for these syndromes and should be prepared to treat the novel adverse events.
References:
- Tran E, Longo DL, Urba WJ. N Engl J Med. 2017;377:2593-6.
- Learn More Here
- Neelapu SS, Tummala S, Kebriaei P, et al. Chimeric antigen receptor T-cell therapy – assessment and management of toxicities. Nat Rev Clin Oncol. 2018;15:47-62.
- Rubin DB, Danish HH, Ali AB, et al. Neurological toxicities associated with chimeric antigen receptor T-cell therapy. Brain. 2019;142:1334-48.
Paul S. Jansson, MD, MS
Emergency physician, critical care fellow, Brigham Health and Harvard Medical School



